 
In chemistry and atomic physics, an electron shell, or principal energy level, may be thought of as the orbit of one or more electrons around an atom's nucleus. The energy spectrum of a system with such discrete energy levels is said to be quantized. The term is commonly used for the energy levels of the electrons in atoms, ions, or molecules, which are bound by the electric field of the nucleus, but can also refer to energy levels of nuclei or vibrational or rotational energy levels in molecules. 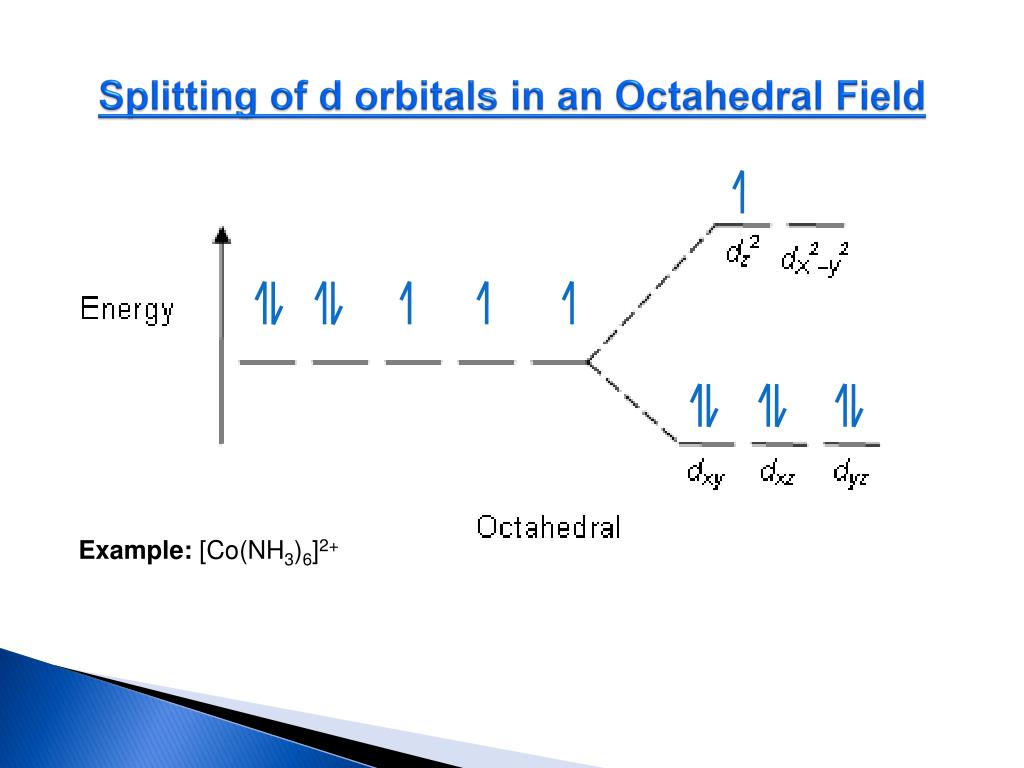
This contrasts with classical particles, which can have any amount of energy. If elements are not aiming for noble gas structures when they form ions, what decides how many electrons are transferred? The answer lies in the energetics of the process by which the compound is made.A quantum mechanical system or particle that is bound-that is, confined spatially-can only take on certain discrete values of energy, called energy levels.  
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
